Naphthenates
Naphthenates are organic carboxylate salt that are formed through interaction of naphthenic acids in crude oil with metal ions such as calcium and sodium in produced water with high pH.
Contents
Conditions for naphtenates to form
The naphthenic acids in crude oil, regardless of their structure, reside harmlessly in the oil phase under reservoir conditions and demonstrate low interfacial activity[1]. As the oil moves out of the reservoir, up through the production tubing and ultimately into the topside separation equipment, depressurization occurs. The depressurization can cause carbon dioxide to flash to the gas phase and the water pH to increase, resulting in naphthenate salts with ions from the water and naphthenic acids from the oil[2].
Production problems caused by naphthenates
Naphthenates are sticky solids, harden on contact with air, and can foul pipelines and processing equipment, causing flow assurance and reduced flow issues and, in worst case scenarios, frequent unplanned shut-downs.
Insoluble in either the oil or water phase, and with a density between that of oil and water, naphthenates tend to accumulate at the oil/water interface and act as surfactants to help stabilize emulsions and form ragged interfaces, with sodium naphthenate being the major contributor for this problem.
It also results in high calcium content in the export crude and causing problems in refineries.
Molecular structure

Naphthenic acid species can be characterized using Gas Chromatography and Mass Spectrometry (GC-MS), two-dimensional GC-MS, Fourier-Transform Ion Cyclotron Resonance (FT-ICR) mass spectrometry and Nuclear Magnetic Resonance (NMR) spectroscopy.
It was traditionally believed that the "damaging" naphthenic acids had molecular weights in the ranges of 200-500. However more recent analysis of naphthenate deposits from a wide range of fields has shown that the main naphthenic acid components are C80 tetra acids containing four to eight cyclic rings, sometimes referred to as ARN acids[3].
These groups afford the molecule unusually high reactivity. The four carboxylic groups tend to create polymeric salt when they are coordinated with divalent metal ions. This weaved polymeric-like structure yields a very sticky deposit that hardens upon contact with air. Once a deposit sets, it can only be dissociated by using a mixture of hot acid and aromatic solvent in laboratory conditions. A remediation using mineral acids in the field would typically be too severe from a safety and corrosion perspective.
Prevention
Acid
The traditional way to avoid naphthenate deposition is to lower the pH by adding an acid. Field experience has shown that lowering the pH to around 6.0 prevents the formation of naphthenate deposits. Further lowering of the pH has no additional benefit and increase corrosion risks[4]. Typical acids that have been used include:
- Inorganic acids, such as phosphoric acid
- Organic acids, such as acetic acid, glycolic acid
- Surfactant acid, such as DoDecylBenzene Sulfonic Acid (DDBSA)
The use of acids for naphthenate control presents logistical issues with regard to safety and storage. Handling corrosive materials in large quantities presents safety issues for the operations personnel. Chemical storage offshore is always at a premium, as the space available is not flexible.
Naphthenate inhibitors
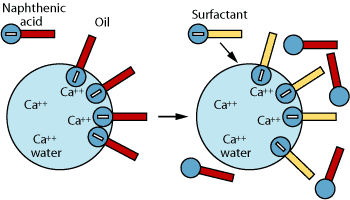
Another method of controlling naphthenate salts is the use of specialty surfactants known as naphthenate inhibitors. These surfactants use the mechanism of interfacial crowding to inhibit salt formation. The surfactant is more interfacially active than naphthenic acid, which reduces the probability of a reaction between the salt and the acid.
Application
Treatment methods and selection of injection points depend on the actual structures of naphthenic acids. For example, the injection point in a fluid with tetrameric naphthenic acid, or ARN, is much more critical than in a fluid with monoacid-type naphthenic acid.
In the case of the ARN acid, the treatment must be upstream of the point where naphthenates are formed because dissociation after the formation is very difficult. The converse is true with a monoacid: After salt formation, dissociation may still be simple, providing some flexibility to the treatment plan. There are case histories that require downhole treatments because of the difficulty of dissociation.
Diagnosis
Until recently, naphthenate salt problems have been dealt with reactively, and not planned for in the design phase. Although it is possible to predict high pH, there are no time-tested early indicators of the presence of the naphthenic acids that would cause a problem.
The problem can be identified in field as the presence of a strong rag layer or by the solubility of an organic deposit. Analytical techniques have evolved to a point where certain naphthenic acids can be identified in problem fields.For example, the ARN acid has been identified at 1,200 amu via mass spectroscopy. The structure has been plotted with the aid of NMR. As more is learned about naphthenate salt problems, it will become possible to better predict and thus plan for such issues.
Reference
- ↑ Ubbels, S. J. and M. Turner, “Diagnosing and preventing naphthenate stabilized emulsions during crude oil processing,” presented at the 6th Petroleum Phase Behaviour and Fouling Conference, Amsterdam, June 19-23, 2005.
- ↑ Arla, D. et al., “Influence of pH and water content on the type and stability of acidic crude oil emulsions,” Energy & Fuels, 21, May 2007, pp. 1337-1342.
- ↑ B.E. Smith, G. Fowler, J. Krane, B. Lutnaes, and S.J. rowland, "Separation and identification of high molecular weight tetra acids responsible for calcium napthenate deposition", 8th International conference on Petroleum Phase Behavior and Fouling, Pau, France, 10-14, 2007
- ↑ M.S. Turner and P.C. Smith, "Control on soap scale formation, including naphthenate soaps - drivers and mitigation", SPE 94339